Overview
Microglia are the brain’s resident immune cells, playing a crucial role in maintaining homeostasis and regulating neural function. In a recent study, Professor Gao Zhihua’s team discovered that the commonly used inhibitor PLX5622, while depleting microglia, can also activate a key receptor—Constitutive Androstane Receptor (CAR)—thereby triggering another regulatory pathway that accelerates the metabolism of exogenous substances such as anesthetics and nicotine. This finding opens new avenues for understanding drug metabolism and addiction mechanisms. Let’s take a closer look.
On February 23, 2026, Professor Gao Zhihua’s team at the Brain-Machine Intelligence National Key Laboratory published a paper online in Neuron, titled “More than microglial depletion: PLX5622 activates the hepatic constitutive androstane receptor to alter anesthesia and addiction.” The study revealed, for the first time, that PLX5622—a widely used CSF1R inhibitor for depleting microglia—exerts effects not only in the central nervous system but also directly activates the liver’s CAR. This significantly enhances the metabolism of anesthetics and addictive substances, affecting both anesthesia sensitivity and addiction-related behaviors. The discovery suggests that previous conclusions about microglial function based solely on PLX5622 experiments may need to be reconsidered.

Microglia: Key Regulators of Neural Function
Microglia are the brain’s primary immune cells, crucial for maintaining homeostasis and regulating neural activity. They modulate neuronal firing, influence synaptic plasticity, and affect neurological and psychiatric conditions. Pharmacologically inhibiting CSF1R to deplete microglia is a widely used method to study their function and explore potential therapeutic strategies for neurodegenerative diseases.
Among these inhibitors, PLX5622 is particularly popular due to its high specificity, oral availability, and ability to cross the blood-brain barrier. It has been used extensively in studies of various neurological disorders and behavioral experiments, uncovering unexpected microglial functions. These effects were traditionally attributed solely to microglial depletion via CSF1R inhibition, though potential off-target effects have not been systematically evaluated.
Building on their previous finding that PLX5622 accelerates mice’s recovery from anesthesia (Cao et al., Current Biology, 2023), the team further explored its effects. Surprisingly, while PLX5622 did not alter loss and recovery of the righting reflex induced by inhaled anesthetics such as isoflurane and sevoflurane, it significantly shortened the duration of anesthesia from injected agents (Figure 1).
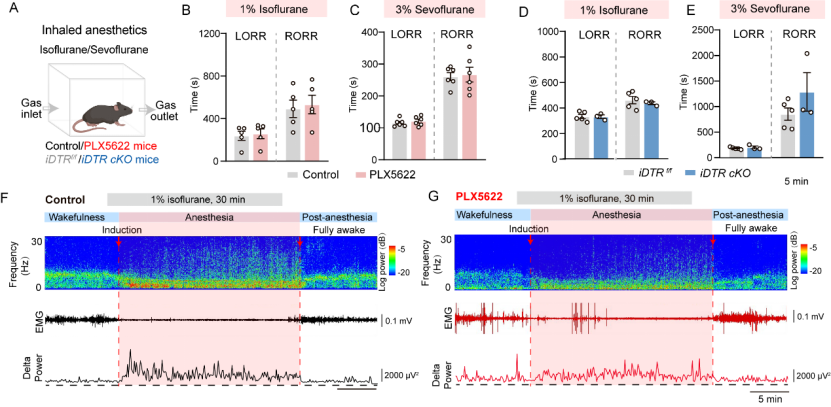
Figure 1. PLX5622 Does Not Affect Inhaled Anesthesia Progression
Why do mice respond differently to injected versus inhaled anesthetics, even though both act on GABAA_AA receptors? The key lies in metabolism: injected anesthetics require hepatic metabolism for clearance, whereas inhaled anesthetics are exhaled without hepatic processing. Mass spectrometry revealed that PLX5622 significantly accelerates barbiturate metabolism. Interestingly, genetic microglial depletion did not produce the same effect, indicating that PLX5622’s impact on hepatic drug metabolism is independent of microglial removal.
PLX5622 Activates Hepatic CAR to Enhance Xenobiotic Metabolism
Cytochrome P450 (CYP) enzymes in the liver are critical for drug metabolism. Transcriptomic analysis of PLX5622-treated livers showed a significant upregulation of enzymes involved in all three stages of xenobiotic metabolism: oxidation, conjugation, and transport (Figure 2). Xenobiotic metabolism is primarily regulated by two hepatic receptors: CAR (NR1I3) and Pregnane X Receptor (PXR, NR1I2), with Cyp2b10 predominantly induced by CAR. Using molecular docking and surface plasmon resonance, the researchers confirmed that PLX5622 binds to and activates CAR, triggering the hepatic xenobiotic metabolism pathway. Knocking out CAR nearly abolished PLX5622-induced hepatic metabolic activation and restored anesthesia duration to baseline.
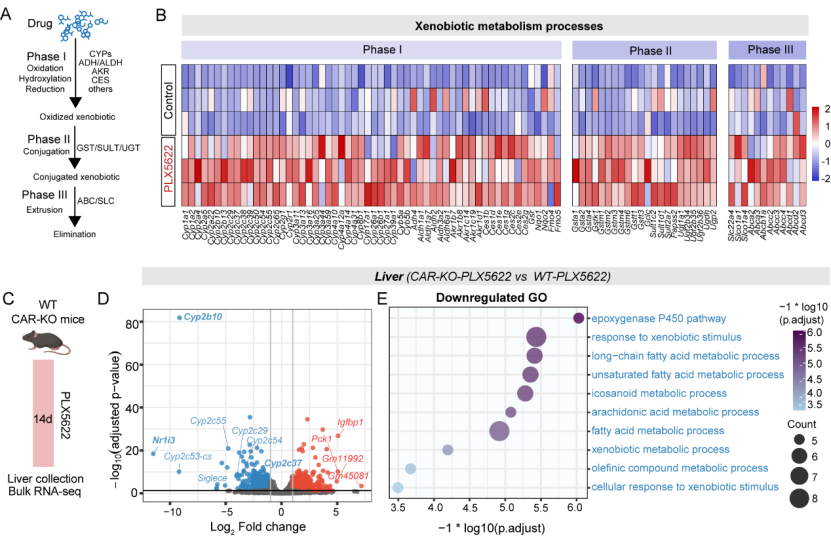
Figure 2. PLX5622 Activates CAR-Dependent Xenobiotic Metabolism
As a potent CAR agonist, PLX5622’s activation of liver enzymes has broad implications for xenobiotic metabolism. Earlier studies that used PLX5622 to deplete microglia in addiction models observed reduced addiction and withdrawal behaviors, previously attributed to microglial loss. Sequencing data now reveal that PLX5622 activates metabolic pathways for nicotine, cocaine, alcohol, and other substances. It accelerates nicotine metabolism and conversion, alleviating withdrawal-related anxiety—a phenomenon absent in CSF1R conditional knockout mice (Cx3Cr1CreER/+::^\text{CreER/+}::CreER/+::CSF1Rfl/fl^\text{fl/fl}fl/fl).
Central-Peripheral “Double Action” of PLX5622
This study is the first to demonstrate that PLX5622 has a dual “central-peripheral” effect: centrally, it inhibits CSF1R in microglia; peripherally, it specifically activates CAR-dependent hepatic xenobiotic metabolism (Figure 3). These findings are a critical reminder for researchers using PLX5622 solely to study microglial function.
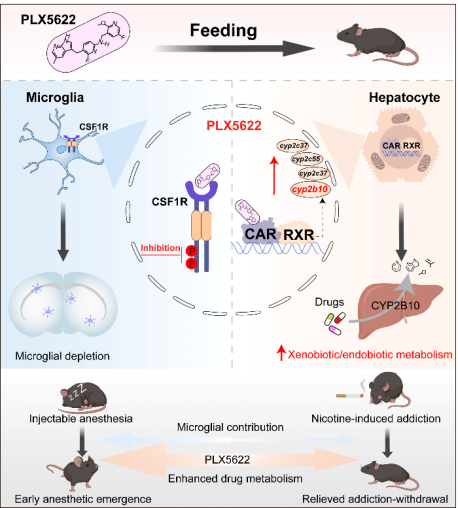
Figure 3. Dual Central and Peripheral Effects of PLX5622
Acknowledgments
The study was led by Professor Gao Zhihua, Brain-Machine Intelligence National Key Laboratory and School of Brain Science and Brain Medicine, Zhejiang University. Dr. Kelei Cao was the first author, with Wangshuo Cheng as co-first author. Additional contributions were made by Liyao Qiu, Ying Yuan, Zexi Wang, Yuqing Zhao, and others. The research was supported by Chief Scientist Shumin Duan, Professors Huan Ma, Zhiying Wu, Tingjun Hou, Cunqi Ye, and Linghui Zeng, among others. Funding came from the National Natural Science Foundation, National Science and Technology Innovation 2030 Major Project, National High-Level Talent Program, Zhejiang Key R&D Projects, Chinese Association for Science and Technology Young Talent Program, and Zhejiang Provincial Natural Science Foundation Exploratory Project.
Read the full paper here:https://doi.org/10.1016/j.neuron.2025.12.044






